
Rotational state, we fall back on Boltzmann's theory:Īn estimation of the distribution is feasible if the distance betweeen the States are in a quite narrow range, mainly the vibrational ground stateĭistribution over J rules the intensity of lines. The population N J and the so-called Hönl-London-FactorĪs the transition probabilities for a simple rotator at high rotational The only occupied vibrational level, the intensity of lines is proportional to Transition and on the occupancy of the involved states. Of course, the intensity of rovibronic lines depends on the probability of a The spin, there is the following approach to the lines of the Q-branch:Īs B v ≈ B v', the lines within this branch lie veryįor the symmetric top, we have the following selection rulesĭiatomic molecules, electronic contribution to J Phenomenona, a detailed analysis is quite complicated. With the NO molecule, we have this situation, but due to spin-related Here, the constant A is in connection with the momentum of electron orbitals.įor Λ ≠ 0, there are new selection rules: Besides ΔJ = ☑, as well 
The respective energy levels are analogous to Molecular total momentum, which includes rotation of nuclei and electrons, the Symbols and selection rules") denotes the projection of this orbital'sĪngular momentum onto the molecule's axis. The quantum number Λ (recall chapter " Term Projection of the electron's rotation onto the axis of The total angular momentum of the molecule. If we consider diatomics, lines of a Q-branch must also have such an angular momentum which contributes to Rotation must shift (ΔK ≠ 0) and a change of the angular momentum of electron As the rotation of the system is maintained, the axis of These lines are connected to transitions with ΔJ = 0 and form the so-called Some molecules display additional lines in the gap between the P- and the R-branch. The shift respective the wavenumbers of the lines of H 35Cl Therefore, two species of HCl molecules with a relativeĭifference of 0.15 percent in their reduced mass are present and some splitting The distance between the first transitions of the two branches isĪlmost all diatomic molecules display such spectra with two branches R and P.Īs an example, hydrogen chloride is shown. The distance Δν between absorption line J and absorption line J+1 becomesįor B v≈ B v' , this distance is approximatelyĢB. Note that B v 0įor ΔJ = - 1 or J - 1 ← J, we speak of absorption peaks within the so-called In turn, the rotational constant Bīecomes dependent on the vibrational state. I v remain for a molecule which has a vibration larger than The third addend and the moment of inertia Within one oscillation, the addend 2 is compensated to Consequently, the moment of inertia becomes I v = µ. Replaced by the average of the square of the the internuclear distance. In the case of a rotator in vibrational motion, r e needs to be Length fixed in the equilibrium position, the moment of inertia is I e Even in such a case the rigid rotor model is a useful point of departure (zeroth-order model).For a diatomic molecule, which we treat as a rigid rotator with a bond Corrections on the rigid model can be made to compensate for small variations in the distance. However, for many actual diatomics this model is too restrictive since distances are usually not completely fixed. The fixed distance between the two masses and the values of the masses are the only characteristics of the rigid model. The linear rigid rotor model consists of two point masses located at fixed distances from their center of mass. More general molecules are 3-dimensional, such as water (asymmetric rotor), ammonia (symmetric rotor), or methane (spherical rotor). A special rigid rotor is the linear rotor requiring only two angles to describe, for example of a diatomic molecule. To orient such an object in space requires three angles, known as Euler angles. 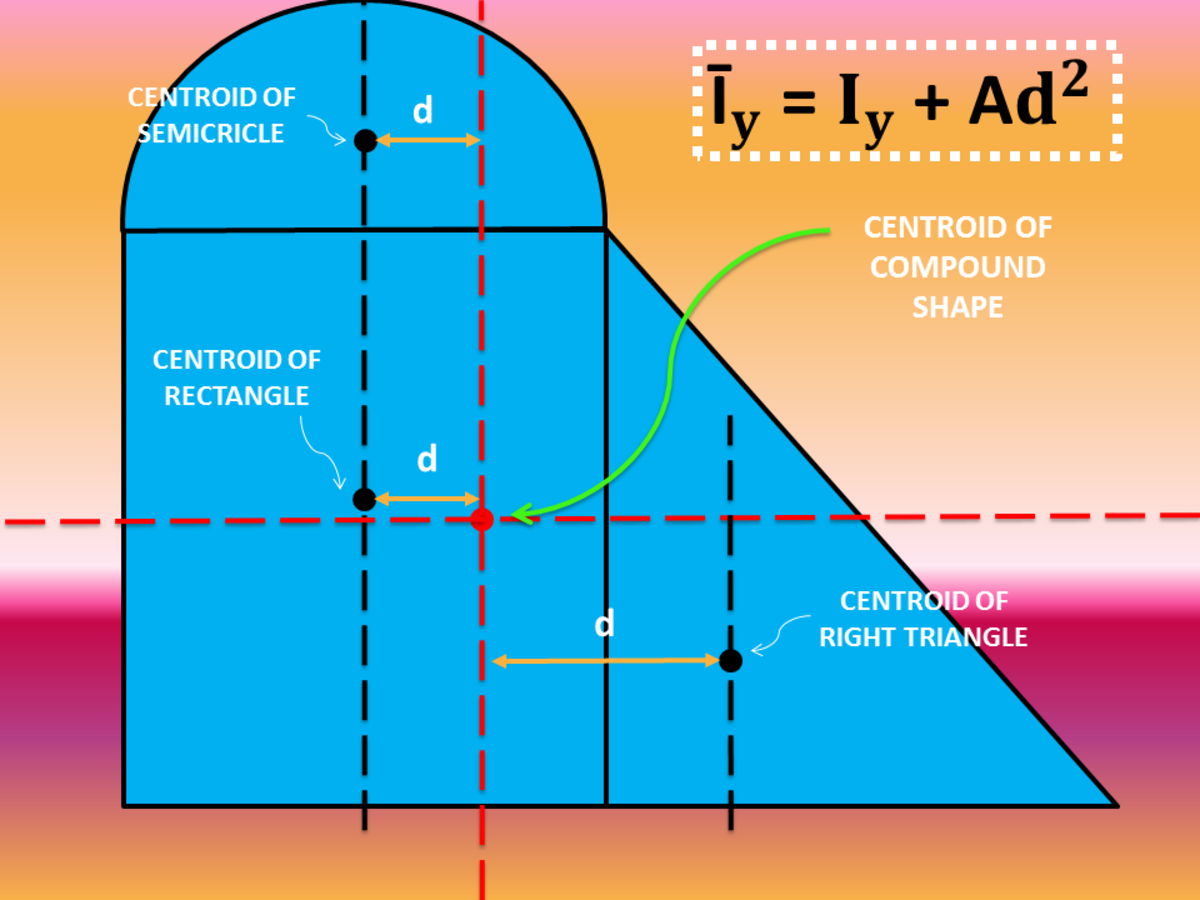
An arbitrary rigid rotor is a 3-dimensional rigid object, such as a top. In rotordynamics, the rigid rotor is a mechanical model of rotating systems. 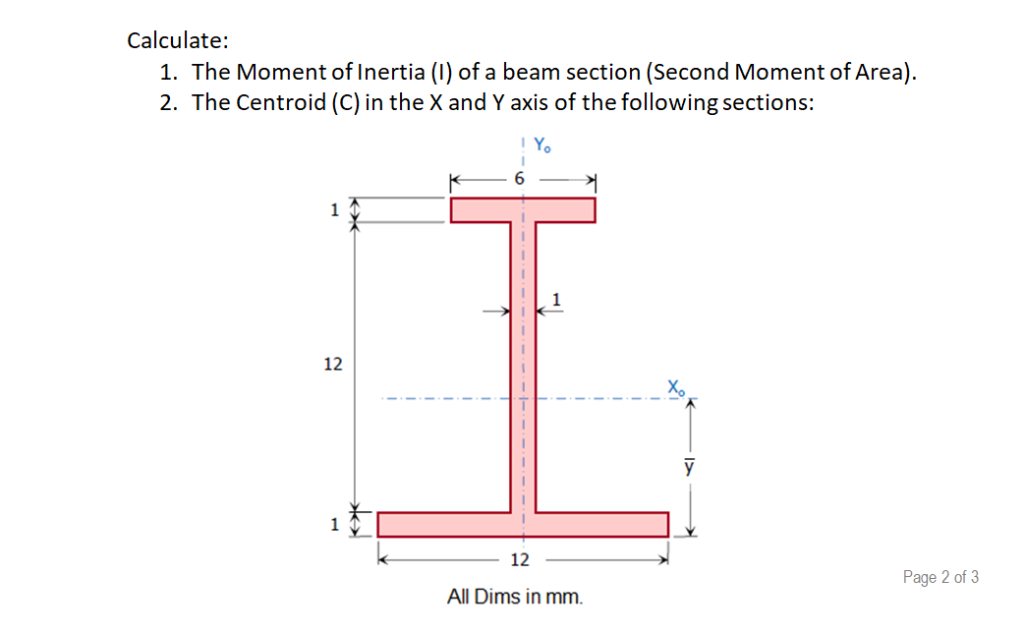
For bond-rotation within a molecule, see conformational isomerism.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
